- Joined
- Apr 9, 2024
- Messages
- 2,028
- Reaction score
- 8,191
Why Route of Administration Doesn’t Reduce Hepatotoxic Risk in 17α-Alkylated Anabolic Steroids
By Type-IIx
Key Takeaways
17α-alkylated steroids are hepatotoxic due to their molecular structure, not administration route.
Sublingual and buccal routes do not meaningfully reduce liver toxicity despite bypassing first-pass metabolism.
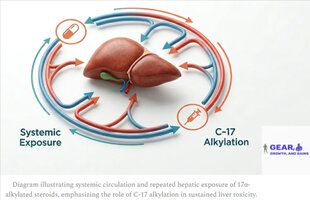
Injectable 17α-alkylated steroids still expose the liver repeatedly via systemic circulation.
Liver damage arises from androgen receptor activation causing mitochondrial ROS, not just liver metabolism.
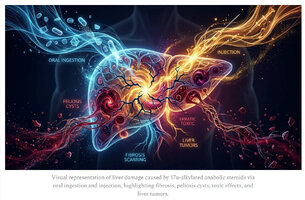
Early symptoms of liver toxicity include fatigue, nausea, and elevated liver enzymes; advanced cases show jaundice and tumors.
Monitoring liver enzymes (ALT, AST, GGT) and clinical signs is essential for all users of 17α-alkylated steroids.
Risk mitigation includes limiting cycle duration, avoiding alcohol, and considering hepatoprotective strategies.
The misconception that injectable 17α-alkylated steroids are “liver-safe” can lead to dangerous dosing practices.
Informed decision-making and education are critical to prevent serious liver complications.
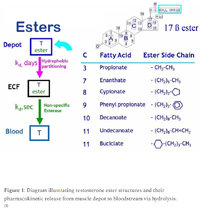
The common wisdom, which is 100% correct and true, is that anabolic-androgenic steroids designed to be taken by intramuscular injection with C-17 esterification – like testosterone (e.g., enanthate), nandrolone (Deca durabolin®, NPP), drostanolone (Masteron), and even trenbolone – are significantly less toxic to the liver (hepatotoxic) than their 17α-alkylated counterparts (17AAs) that are designed to be taken by mouth, like methyltestosterone (METHITEST™, norethandrolone (Nilevar®), methasterone (Superdrol), and mibolerone (R1881; methyltrienolone). [1] C-17 esterification is a chemical modification where an ester like enanthate or decanoate is attached to the steroid to increase its half-life by attaching a carbon chain in the 17-β orientation. [1-1] This modification effectively increases the steroid’s half-life by increasing the residence time inside the depot that is formed in the muscle tissue, from where it slowly dissipates into the blood circulation, where esterases actively hydrolyse the molecule, breaking down those fatty acid bonds of the carbon chain, activating the parent hormone, whose biological half-life is much shorter than the prohormone’s. [1-2] [2]
Esters: Fatty acid bonds of different aliphatic or other carbon chain lengths attached to the steroid’s 17β-hydroxyl group to produce a prodrug for oil vehicle that when injected deep into the belly of muscle is released from depot at a rate determined by its partitioning coefficient according to the ester’s hydrophobicity or water-solubility before entering the extracellular fluid of whole blood where it is rapidly hydrolyzed by esterase…
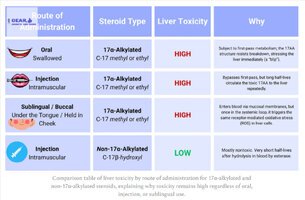
The myth that many bodybuilders mistakenly embrace, though, is a belief that that this hepatotoxicity is dictated by how they’re taken. By corollary, these same bodybuilders might mistakenly believe that sublingual (or buccal) ingestion of 17AA meaningfully improves their otherwise toxic effects because it bypasses first pass metabolism by the liver.
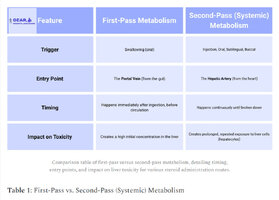
First Pass vs. Second Pass Metabolism
First pass metabolism refers to the process by which a drug is metabolized by the liver before it enters systemic circulation. This can result in a decreased concentration of the drug reaching the bloodstream, as some of it is metabolized and excreted before reaching the intended target site. [4]
Second pass metabolism, on the other hand, refers to the metabolism of a drug that has already entered systemic circulation and is circulating throughout the body. This can occur in various tissues and organs in addition to the liver. [4-1]
Why Are 17AAs So Hard on the Liver?
So what distinguishes oral 17AAs from the esterified steroids? Oral bioavailability, of course. Why are they bioavailable but non-17AAs are not? Oral delivery of testosterone, for example – just swallowing, say, testosterone enanthate – would be followed by rapid metabolism in the liver to such an extent that only the earliest phases of its metabolism would occur: it is deactivated and cannot survive delivery to muscle tissue. [4-2]
FAQ
People Also Ask
Q: Does injecting 17α-alkylated steroids eliminate the risk of liver damage?
A: No. Injectable 17α-alkylated steroids bypass first-pass metabolism but still expose the liver repeatedly through systemic circulation. The hepatotoxic risk remains high due to the steroid’s chemical structure and prolonged half-life.
The reason that 17AAs are so hepatotoxic is because C-17 alkylation prolongs their half-lives and increases their potency (e.g., Superdrol is methyl-Masteron), from mere seconds to hours or even days, or large parts thereof. According to The Bond and Llewellyn Hypothesis About Anabolic Androgenic Steroid-Induced Hepatotoxicity, a steroid’s hepatotoxicity is described by the Equation:
Equation 1: Hepatotoxicity is the product of half-life (t1/2) and potency to activate the androgen receptor (AR) [5]
All anabolic steroids, including testosterone, are toxic. How do we know? Because even testosterone produces endogenous metabolites (e.g., epitestosterone, androsterone, etiocholanolone) meaning that it must be metabolized or broken down in phases through firstand secondpass metabolism by the liver to more hydrophilic molecules in order for the body to excrete them. [4-3]
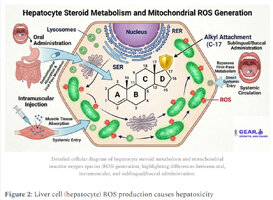
Anabolic steroids increase reactive oxygen species (ROS) in tissues that include liver and cells that include the basic unit of the liver, hepatocytes. [5-1] This is really where it all goes down, and the aminotransferases like ALT and AST start to take a hit and ratchet up accordingly.
This guide will examine the scientific evidence behind steroid-induced liver damage, exploring why the route of administration doesn’t meaningfully alter the hepatotoxic risk profile of 17α-alkylated steroids.
The Science Behind the Myth
Take the example of injectable Superdrol (methasterone; 17α- methyldrostanolone), a popular black market methasterone formulation in oil vehicle. This preparation is in demand because of this myth that’s promulgated by marketing to this very misconception: that injecting Superdrol is non-liver toxic.
Unfortunately, while it is true that injectable administration bypasses first-pass metabolism, this has scant practical significance since the damage that’s done by methasterone arises from its half-life (t1/2 ) and potency to activate the AR, which is on the one hand protracted by 17α-alkylation (17-CH3-) and on the other hand potentiated by the same. Masteron is fairly weak and it’d be broken down very quickly if just taken by mouth, like testosterone. Not so with Superdrol… quite the contrary, in fact.
The Bond and Llewellyn Hypothesis About Anabolic Androgenic Steroid-Induced Hepatotoxicity suggests that hepatotoxicity results from androgen receptor activation and subsequent mitochondrial dysfunction, not from the liver’s attempt to metabolize these compounds. This mechanistic understanding explains why bypassing first-pass metabolism doesn’t provide meaningful protection.
FAQ
People Also Ask
Q: What are early signs of steroid-induced liver toxicity?
A: Early symptoms include fatigue, nausea, loss of appetite, and abdominal discomfort. Elevated liver enzymes (ALT, AST) are common biochemical markers. More severe signs include jaundice, dark urine, and light-colored stools.
The Bond and Llewellyn Hypothesis About Anabolic-Androgenic Steroid Hepatotoxicity
The 2016 Medical Hypothesis proposed by Bond and Llewellyn offers a nuanced explanation of how 17α-alkylated steroids induce hepatotoxicity. Their hypothesis centers on the increase of ROS, these reactive oxygen species, through carnitine palmitoyltransferase 1 (CPT1) activity and mitochondrial fatty acid β-oxidation. [5-2]
By Type-IIx

Key Takeaways
17α-alkylated steroids are hepatotoxic due to their molecular structure, not administration route.
Sublingual and buccal routes do not meaningfully reduce liver toxicity despite bypassing first-pass metabolism.
Injectable 17α-alkylated steroids still expose the liver repeatedly via systemic circulation.
Liver damage arises from androgen receptor activation causing mitochondrial ROS, not just liver metabolism.
Early symptoms of liver toxicity include fatigue, nausea, and elevated liver enzymes; advanced cases show jaundice and tumors.
Monitoring liver enzymes (ALT, AST, GGT) and clinical signs is essential for all users of 17α-alkylated steroids.
Risk mitigation includes limiting cycle duration, avoiding alcohol, and considering hepatoprotective strategies.
The misconception that injectable 17α-alkylated steroids are “liver-safe” can lead to dangerous dosing practices.
Informed decision-making and education are critical to prevent serious liver complications.
The common wisdom, which is 100% correct and true, is that anabolic-androgenic steroids designed to be taken by intramuscular injection with C-17 esterification – like testosterone (e.g., enanthate), nandrolone (Deca durabolin®, NPP), drostanolone (Masteron), and even trenbolone – are significantly less toxic to the liver (hepatotoxic) than their 17α-alkylated counterparts (17AAs) that are designed to be taken by mouth, like methyltestosterone (METHITEST™, norethandrolone (Nilevar®), methasterone (Superdrol), and mibolerone (R1881; methyltrienolone). [1] C-17 esterification is a chemical modification where an ester like enanthate or decanoate is attached to the steroid to increase its half-life by attaching a carbon chain in the 17-β orientation. [1-1] This modification effectively increases the steroid’s half-life by increasing the residence time inside the depot that is formed in the muscle tissue, from where it slowly dissipates into the blood circulation, where esterases actively hydrolyse the molecule, breaking down those fatty acid bonds of the carbon chain, activating the parent hormone, whose biological half-life is much shorter than the prohormone’s. [1-2] [2]
Esters: Fatty acid bonds of different aliphatic or other carbon chain lengths attached to the steroid’s 17β-hydroxyl group to produce a prodrug for oil vehicle that when injected deep into the belly of muscle is released from depot at a rate determined by its partitioning coefficient according to the ester’s hydrophobicity or water-solubility before entering the extracellular fluid of whole blood where it is rapidly hydrolyzed by esterase…

The myth that many bodybuilders mistakenly embrace, though, is a belief that that this hepatotoxicity is dictated by how they’re taken. By corollary, these same bodybuilders might mistakenly believe that sublingual (or buccal) ingestion of 17AA meaningfully improves their otherwise toxic effects because it bypasses first pass metabolism by the liver.
First Pass vs. Second Pass Metabolism
First pass metabolism refers to the process by which a drug is metabolized by the liver before it enters systemic circulation. This can result in a decreased concentration of the drug reaching the bloodstream, as some of it is metabolized and excreted before reaching the intended target site. [4]
Second pass metabolism, on the other hand, refers to the metabolism of a drug that has already entered systemic circulation and is circulating throughout the body. This can occur in various tissues and organs in addition to the liver. [4-1]

Why Are 17AAs So Hard on the Liver?
So what distinguishes oral 17AAs from the esterified steroids? Oral bioavailability, of course. Why are they bioavailable but non-17AAs are not? Oral delivery of testosterone, for example – just swallowing, say, testosterone enanthate – would be followed by rapid metabolism in the liver to such an extent that only the earliest phases of its metabolism would occur: it is deactivated and cannot survive delivery to muscle tissue. [4-2]
FAQ
People Also Ask
Q: Does injecting 17α-alkylated steroids eliminate the risk of liver damage?
A: No. Injectable 17α-alkylated steroids bypass first-pass metabolism but still expose the liver repeatedly through systemic circulation. The hepatotoxic risk remains high due to the steroid’s chemical structure and prolonged half-life.
The reason that 17AAs are so hepatotoxic is because C-17 alkylation prolongs their half-lives and increases their potency (e.g., Superdrol is methyl-Masteron), from mere seconds to hours or even days, or large parts thereof. According to The Bond and Llewellyn Hypothesis About Anabolic Androgenic Steroid-Induced Hepatotoxicity, a steroid’s hepatotoxicity is described by the Equation:
Equation 1: Hepatotoxicity is the product of half-life (t1/2) and potency to activate the androgen receptor (AR) [5]
All anabolic steroids, including testosterone, are toxic. How do we know? Because even testosterone produces endogenous metabolites (e.g., epitestosterone, androsterone, etiocholanolone) meaning that it must be metabolized or broken down in phases through firstand secondpass metabolism by the liver to more hydrophilic molecules in order for the body to excrete them. [4-3]
Anabolic steroids increase reactive oxygen species (ROS) in tissues that include liver and cells that include the basic unit of the liver, hepatocytes. [5-1] This is really where it all goes down, and the aminotransferases like ALT and AST start to take a hit and ratchet up accordingly.
This guide will examine the scientific evidence behind steroid-induced liver damage, exploring why the route of administration doesn’t meaningfully alter the hepatotoxic risk profile of 17α-alkylated steroids.
The Science Behind the Myth
Take the example of injectable Superdrol (methasterone; 17α- methyldrostanolone), a popular black market methasterone formulation in oil vehicle. This preparation is in demand because of this myth that’s promulgated by marketing to this very misconception: that injecting Superdrol is non-liver toxic.
Unfortunately, while it is true that injectable administration bypasses first-pass metabolism, this has scant practical significance since the damage that’s done by methasterone arises from its half-life (t1/2 ) and potency to activate the AR, which is on the one hand protracted by 17α-alkylation (17-CH3-) and on the other hand potentiated by the same. Masteron is fairly weak and it’d be broken down very quickly if just taken by mouth, like testosterone. Not so with Superdrol… quite the contrary, in fact.
The Bond and Llewellyn Hypothesis About Anabolic Androgenic Steroid-Induced Hepatotoxicity suggests that hepatotoxicity results from androgen receptor activation and subsequent mitochondrial dysfunction, not from the liver’s attempt to metabolize these compounds. This mechanistic understanding explains why bypassing first-pass metabolism doesn’t provide meaningful protection.
FAQ
People Also Ask
Q: What are early signs of steroid-induced liver toxicity?
A: Early symptoms include fatigue, nausea, loss of appetite, and abdominal discomfort. Elevated liver enzymes (ALT, AST) are common biochemical markers. More severe signs include jaundice, dark urine, and light-colored stools.
The Bond and Llewellyn Hypothesis About Anabolic-Androgenic Steroid Hepatotoxicity
The 2016 Medical Hypothesis proposed by Bond and Llewellyn offers a nuanced explanation of how 17α-alkylated steroids induce hepatotoxicity. Their hypothesis centers on the increase of ROS, these reactive oxygen species, through carnitine palmitoyltransferase 1 (CPT1) activity and mitochondrial fatty acid β-oxidation. [5-2]